
1 / 5










| Product Code | Product Name | Model | Technical Description |
|---|---|---|---|
| 030020003 | Kyphoplasty Tool Kit | Type B (Lumbar) | φ3.0 Access Device (1pc), φ3.5 Aiguille (1pc), Guide Wire (1pc), φ4.2 Expansion Cannula (1pc), φ3.5 Cement Applier (4pc) |
| 030020002 | Kyphoplasty Tool Kit | Type D (Lumbar) | φ3.0 Access Device (2pc), φ3.5 Aiguille (1pc), Guide Wire (2pc), φ4.2 Expansion Cannula (2pc), φ3.5 Cement Applier (6pc) |
| 030020001 | Kyphoplasty Tool Kit | Type G (Lumbar) | φ4.2 Access Device (1pc), φ3.5 Aiguille (1pc), Guide Wire (1pc), φ3.5 Cement Applier (4pc) |
| 030030005 | Kyphoplasty Tool Kit | Type T1 (Thoracic) | φ2.4 Access Device (2pc), φ3.0 Aiguille (1pc), Guide Wire (2pc), φ3.6 Expansion Cannula (2pc), φ3.0 Cement Applier (6pc) |
| 030030006 | Kyphoplasty Tool Kit | Type T11 (Thoracic) | φ2.4 Access Device (1pc), φ3.0 Aiguille (1pc), Guide Wire (1pc), φ3.6 Expansion Cannula (1pc), φ3.0 Cement Applier (4pc) |
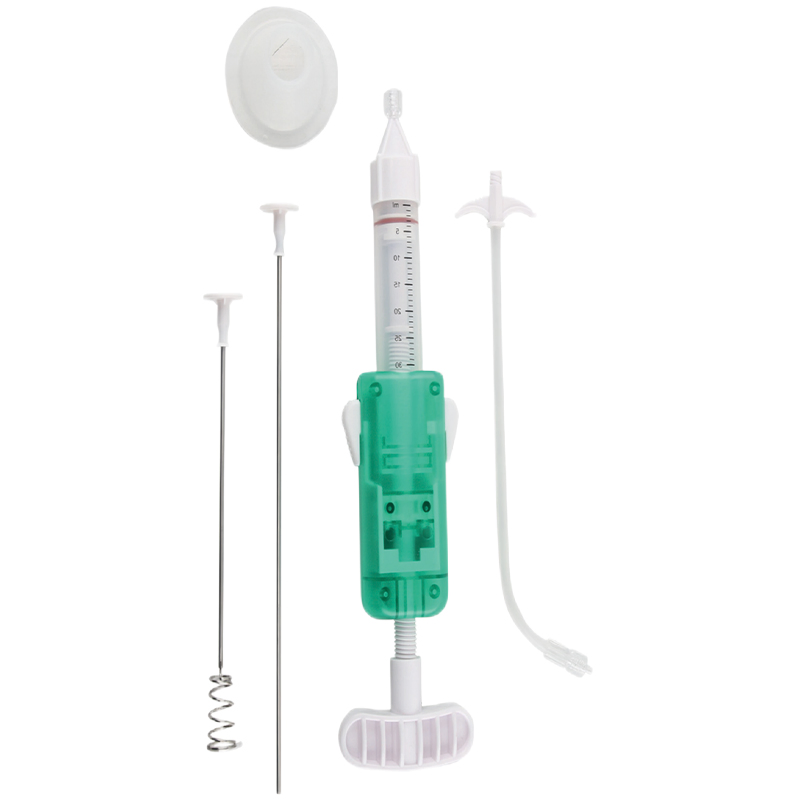
Our integrated design allows for quick and efficient percutaneous access to the bone, creating a bone tissue guide channel seamlessly. This significantly reduces trauma for the patient. We offer both bevel and diamond tips, allowing medical professionals to select the best option based on specific clinical needs.
*Note: Prices are for reference only. Final pricing is subject to the latest official quotation.